A Technical Overview by Dr. Caibin Xiao
The concepts of free chlorine and total chlorine are misleading and the cause for significant con-fusion in the water treatment industry. In this short paper, we would like to provide an overview of the underlying chemistry to understand the use of the terms related to the measurement and control of chlorine in a real-world process.
Sodium Hypochlorite as the Chlorine Source
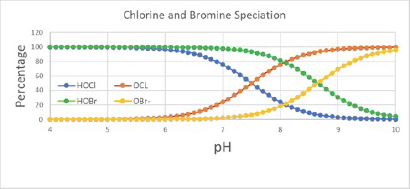
Chlorine exists as hypochlorous acid and hypochlorite ion in waters that do not contain any organic or inorganic nitrogen compounds. The ratio of these two species is dependent on the water pH and temperature. Above pH 8.5, hypochlorite ion is the dominant species.
HCl(aq) ← > H + (aq) + ClO – (aq)
The free chlorine concentration is the stoichiometric total of these two species regardless of the water pH and temperature. Some people may think that free chlorine should only include hypo-chlorous acid because the biocidal power of hypochlorite ion is believed to be less than hypochlor-ous acid.
Monochloramine
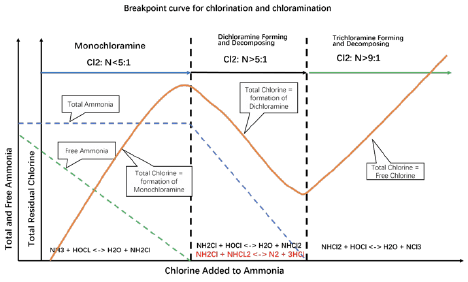
Sodium hypochlorite reacts with ammonium ions rapidly in the pH range common in the municipal and industrial waters. When the chlorine to ammonium molar ratio is less than 1:1, or the chlorine to nitrogen ppm ratio (Cl2/N, ppm/ppm) is less than 5:1, monochloramine, NH2Cl, is the only chlo-ramine species that is produced. Monochloramine is a chemically well-defined species, although its pure form is not stable to be separated.
Beyond Monochlormaine & the Breakpoint
When the chlorine to ammonium ratio is greater than 1, dichloramine starts to form. This is com-monly referred to as the breakpoint chlorination process. As dichloramine is forming, a series of re-actions happen leading to dichloramine decomposition to nitrogen gas and non-oxidizing chloride. This is the reason why the breakpoint curve has a dip – as more chlorine is pumped to a system, the monitored ORP or total chlorine declines rather than increase. This decline is not an artifact of any measurement method used. It is a real decrease of the total oxidizing species due to the decompo-sition reaction below. When this happens in a water stream containing above 10 ppm total chlorine, nitrogen bubbles are generated and visible to the naked eye.
NH2Cl + NHCl2 → N2 + 3HCl
DPD Free & Total Methods
DPD stands for N,N-diethyl-p-phenylenediamine. It reacts with chlorine to form a red colored spe-cies, at pH between 5.0 – 6.5, that can be analyzed with a spectrophotometer or a LED-based colorimeter at a wavelength around 560nm. The DPD reagent containing the necessary buffer can be formulated as a single solid mixture or a liquid. The liquid reagent is less stable than the solid mixture. For inline applications, two DPD reagents are used. The DPD containing the liquid reagent is acidified to very low pH to prevent DPD from being oxidized by oxygen. The second liquid reagent is a buffer. The free chlorine concentration determined by the free DPD method is the stoichiometric total of both hypochlorous acid and hypochlorite ion regardless of the sample pH.
The DPD free chlorine method can determine as low as 0.01 ppm chlorine. The challenge for ultra-low range chlorine analysis is variation caused by the reagent color background.
Total chlorine is the total account of free chlorine and all chloramines, including monochloramine, dichloramine, and other organic chloramines. DPD can react with iodide in the same wayas the chlorine iodide reaction. Most chloramines can react with iodide to form iodine. The total DPD reagent pack contains iodide to determine the total concentration of chloramines including monochloramine and dichloramine via iodine chemistry.
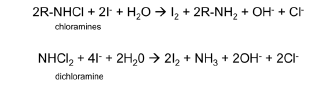
DPD without iodide also reacts slowly with some chloramines including monochloramine, especial-ly when the sample temperature is above 70 Fahrenheit (or 21 ºC). For this reason, the DPD free method is not entirely selective to only free chlorine. If the chloramine concentration is high, one can easily have a detectable amount of free chlorine with the free chlorine reagent. DPD can be oxidized by almost all commonly used oxidizing biocides and other oxidizers such as oxygen and
MnO2.
The Indophenol Method for Monochloramine
The indophenol method for monochloramine is a monochloramine specific colorimetric method. The reagent selectively reacts with monochloramine to form a green-color indophenol. If the water does not contain other chloramines, the total DPD chlorine concentration should agree with the concentration by the indophenol method. Since the total DPD method can pick up dichloramine and the indophenol does not, the total chlorine concentration will be greater than the monochloramine concentration if beyond breakpoint chlorination occurs and dichloramine starts to form.
Another way to detect the onset of dichloramine formation is to test free chlorine in the monochlo-ramine treated water. The DPD free chlorine reagent does not react with monochloramine in a significant amount at sample temperature below 80 ºF within 60 seconds. For municipal waters treated with monochloramine, less than 0.1 ppm chlorine is commonly determined by the free DPD reagent. If the free chlorine concentration determined by the free DPD reagent is above 0.1 ppm, it could be an indication of dichloramine formation. In a well-treated monochloramine water, total chlorine concentration should be in close agreement with the monochloramine concentration, and the free chlorine concentration should be less than 0.1 ppm. Adding to the confusion, when we talk about ppm monochloramine we are talking about ppm monochloramine as ppm chlorine rather than ppm NH2Cl.
Industrial Cooling Water
Sodium hypochlorite and, less commonly, chlorine gas are used for biological growth control in the industrial cooling water systems. Inevitably, cooling water in the cooling systems contains intentionally added nitrogen containing treatment chemicals, such as azoles, and natural occur-ring organic nitrogen containing compounds from the makeup. For some cooling systems, process chemicals leaking into the cooling water is another source of nitrogen containing contaminants. Thus, the cooling water contains a range of oxidative chlorine species, including free chlorine and chloramines.
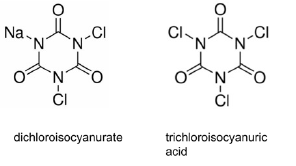
Tri & Di-Chloroisocyanuric Acid
Trichlor and dichlor isocyanurate are chloramines. Through a series of hydrolysis reactions when these chlorine precursors are dissolved in water, more than 10 oxidative species including hypo-chlorous acid and hypochlorite are formed. The free chlorine to the total chlorine ratio is a function of cyanurate concentration, pH, and the system demand on chlorine. The free chlorine DPD method may not be a consistent way to monitor the chlorine dose. Instead, the total chlorine DPD method offers more consistency.
Bromine & Bromine Precursors
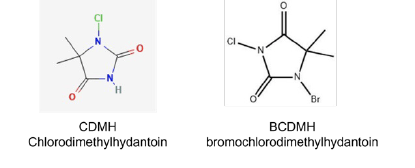
Many small and medium sized cooling towers are treated with bromine. Bromine is added to the cooling water system either from a bromine precursor such BCDMH (bromochlorodimethylhydantoin) or a sulfamate-stabilized liquid bromine formulation. These bromine precursors are bromoamines by definition. They hydrolyze in water to form free hypobromite. In water without demand, a molar BCDMH can produce one molar bromine and one molar chlorine. The latter hydrolyzing reaction is slow. The DPD free method will pick up the bromine portion while the total method will pick up the total including the contribution from CDMH.
It is unclear if the bromine precursors completely hydrolyze to form hypobromous acid and hypo-bromite. Bromoamines are more reactive toward DPD. The DPD free method will certainly detect portion of total bromoamine while the total DPD method will detect the total concentration includ-ing hypobromous acid, hypobromite, and unhydrolzed bromine-sulfamate. For dosing control pur-poses, either free bromine by the free DPD method or the total DPD method can be used. Like the situation for chlorinated systems with heavy nitrogen containing contaminants, the total bromine by the total DPD method should be periodically checked to prevent excess amount of bromoam-ines.

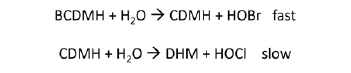
Methods to Measure Free-Total Chlorine & Monochloramine other than DPD
Methods for free chlorine analysis can be roughly categorized into three:
- Regeaent based spectrophotometric methods. DPD is the most widely used reagent for the spectrophotometric determination of chlorine.
- Direct spectrophotometric method. Hypochlorous acid has a distinct absorption band around 280 nm. The monochloramine band is at 245 nm while the dichloramine has a strong band at 203 nm and a weak band at 290 nm. These UV absorption bands can be used for the direct spectro-photometric determination of free, monochloramine and dichloramine. It is especially useful to simultaneously monitor both monochloramine and dichloramine in monochloramine generators.
- Electrochemical based methods.
The electrochemical method is based on the property that hypochlorous acid can be reduced at gold or platinum electrode. The sensors for chlorine can be further divided into two categories. The membrane based amperometry sensors have a porous membrane separating the sensing electrode and the inner electrolyte from the sample water containing chlorine to be determined. The bare gold type of amperometric sensors do not have the inner electrolyte. The sensing electrode is in direct contact with the chlorine species to be determined in the water sample.
In membrane based amperometric sensing, the sensor signal is proportional to the diffusion rates of hypochlorous acid and hypochlorite ions through the sensor membrane. The diffusion rates of these two ions are quite different. Thus, the sensor signal is a function of the sample pH. To report the free chlorine concentration as the total of hypochlorous acid and hypochlorite ion, comparable with the DPD free method, a pH compensation must be implemented in the aperometric sensor.
In non-membrane amperometric sensing, the sensor signal is proportional to the diffusion rates of hypochlorous acid and hypochlorite ions from the bulk water directly to the sensor electrode surface. Like the membrane-based sensor, a pH compensation is required for the free chlorine concentration.
Membrane based amperometric sensors offer some selectivity towards different oxidative species by selecting the membrane material and inner electrolyte. This type of sensor cannot distinguish free chlorine from chloramines and will detect a mix signal reflecting the compound contribution from all oxidizing species that can penetrate the sensor membrane. Since the sensor has a different sensitivity to different chloramine agents, the compound signal may not be equivalent to the total chlorine concentration determined by the total DPD method. The total chlorine sensor can be only used in the system with one chloramine, or the system is constant with regarding the chloramine speciation. In this situation whether the sensor is free or total chlorine depends on the concentration value being calibrated against.
The membrane less bare gold amperometric sensor does not rely on the membrane diffusion mechanism. Monochloramine and other chloramines exhibit a small but significant amount of response on the electrode. This is beneficious for obtaining a signal closer to the DPD total for the compli-cated industrial system. If properly calibrated, the bare gold electrode sensors can be ideal for the monitoring and dosing control of chlorine and bromine.
What Should we Measure, Free or Total?
To answer this question, we must understand our goal of measuring chlorine. We want to measure chlorine for the purpose of disinfection. For drinking water, national governments have a chlorine range as a requirement. For industrial systems, guidelines from a variety of organizations or local experience have chlorine targets. A source of confusion is that free chlorine is believed to be a more affective biocide than chloramines. For this belief, people want to achieve a higher measurable free chlorine as the target rather than a reasonable total chlorine target.
For municipal drinking water treated with sodium hypochlorite or chlorine gas, we just need to measure free chlorine. The DPD colorimetric method is a government accepted standard method. Inline amperometric sensors can be calibrated to the free chlorine value measured by the DPD method. Largely, the chlorine concentrations measured by the DPD free method and total method are in close agreement. Some water may exhibit slightly higher chlorine value by the total method.
For industrial systems containing a range of oxidizing species, one can measure either free or total chlorine as the control target. However, if free chlorine is selected to be a routine monitoring tar-get, total chlorine must be measured periodically to access the system total demand to avoid two following scenarios.
- Dosing more and getting less. Like the ammonium – chlorine breakpoint phenomenon, the presence of other organic or inorganic nitrogen compounds in the industrial water could make chlorination process complicated. After all nitrogen containing compounds being converted to mono-chloramines, further adding chlorine will result in a decrease in total chlorine or net oxidizing power loss. By comparing the relative trend and ratio of free chlorine and total chlorine concentrations, over chlorination could be avoided.
- To achieve a given amount of free chlorine results in an excess amount of total chloramine. Systems containing a large amount of nitrogen containing compounds can have a large amount of chlorine demand. If the chlorine control target is solely based on the free chlorine concentration, a large amount of chlorine will be consumed to form chloramines to achieve the desired free target. For this type of system, the fundamental question is whether chloramines are effective for the purpose of disinfection and how much chloramines as total chlorine is needed to achieve the disinfection goal. I have encountered cooling systems that reached 10 ppm total chlorine to have 0.5 ppm free by the free DPD method. The excess amount of chlorine dosed does not only result in wasting chemicals but also causing an increasing amount of corrosion.
In many large municipal water systems, monochloramine is the only chlorine species. Little free chlorine (less than 0.05 ppm) is detected with the free DPD method. The concentration of monochloramine determined by the total DPD should agree with that determined by the monochloramine specific method that is based on the reaction of monochloramine with indophenol to form a blue compound. If the concentration by the total DPD method is greater than that by the indophenol method, it is likely that the water sample contains both monochloramine and dichloramine. Dichloramine can be detected by the iodide containing total DPD method. The discrepancy between these concentrations is an indication of the chlorination approaching the breakpoint.
An Example
A Pyxis Lab® OXIPANEL™ IK-765SS-B Series self-brushing panel containing the Pyxis Lab® ST-765SS series bare-gold sensor was installed in a data center cooling tower. The tower was treated with stabilized bromine. The following chart is a comparison of the sensor reading with offline DPD total values. Since 2019, the ST-765SS Series bare-gold sensor platform has been deployed globally for a wide variety of different applications, including municipal water treated with free chlorine, chlorine dioxide, and monochloramine, simultaneously measuring sulfite and chlorine for RO feed-water and wastewater effluent dichlorination and ozone on bottling water process.
Conclusion
For complicated industrial waters, both free chlorine and total chlorine are empirical terms. The free DPD method can pick up contribution non-free chlorine oxidizers. The total DPD method mea-sured all oxidizers including chloramines that can convert iodide to iodine. Because different chloramines have different reaction kinetics and reactivity to iodide, the total chlorine certainly does not account for all chloramines stoichiometrically. For dosing control, one could choose either free or total as the primary control target and check the second periodically to prevent grossly over or under biocidal feeding. The bare gold electrode sensor measures a mix of signals from all oxidizers that can be electrochemically reducing on the gold electrode, with appropriate calibration to either free or total DPD chlorine, the sensor can be a reliable way for monitoring and dosing oxidizing biocide to a range of applications including industrial cooling towers.



